1WorldSync pilot programme demonstrates process efficiencies that will benefit the global healthcare industry.
PHOENIX group becomes FMD compliant with Movilitas
Falsified medicines pose a serious health risk to patients. The European Falsified Medicines Directive (FMD), which had to be implemented by February 2019, aims to eliminate this risk. The directive demands a secured pharmaceutical supply chain through end-to-end verification of prescribed pharmaceutical products, thereby increasing the safety for patients. All supply chain partners have to fulfill specific requirements to reach compliance. PHOENIX group, a leading healthcare provider in Europe, has been FMD-compliant since April 20th, 2018. The company has chosen the Movilitas. Cloud solution to implement the FMD requirements in 17 countries for wholesale and pre-wholesale activities.
What requirements do stakeholders need to fulfill to comply?
FMD works with basically two verification systems: the central system, which is referred to as the European Medicines Verification System (EMVS), communicates with separate country-based systems for each EU Member State, which are referred to as the National Medicines Verification Systems (NMVSs). FMD mainly involves four different stakeholders: manufacturers, wholesalers, hospital pharmacies, and retail pharmacies.
Manufacturers
Life science manufacturers or Marketing Authorisation Holders (MAH) need to upload the relevant commissioned product data to the EMVS, which will then communicate the data to the National Systems.
Wholesalers
Wholesalers and 3PLs (pre-wholesalers) generally have the same obligations as manufacturers, except that they do not place any products in the market. They have to secure the supply chain and verify any suspicion of falsified medicines. This boils down to authenticating products that are returned through verification in the National System or deactivating/re-activating the product serial number in the National Systems with a reason code.
Hospital pharmacies
EU FMD is also mandatory for hospital pharmacies, to prevent the distribution of counterfeit medicines to patients. Hospitals therefore also need to authenticate and deactivate medicines before dispensing them to the patients (or re-activate them in certain cases) and use the National Systems for this. Besides its requirements, FMD also comes with a number of potential opportunities for hospital pharmacies: reduction in wasted medicines, in expired medicines, in local medicines shortages, as well as increased opportunities to engage in patient care, more rapid product recalls, better inventory management, and improvement in the pharmacy workflow.
Retail pharmacies
In the end, the whole purpose of the EU FMD is to protect the patient and improve patient safety by securing the supply chain. Before dispensing a medicine to a patient, it must be authenticated by scanning and verification against the respective National System. Once a pharmaceutical product has been dispensed, it is de-activated in the system.

Figure 1 FMD Stakeholders
How PHOENIX group fulfilled its FMD requirements in 17 countries for wholesale and pre-wholesale activities
PHOENIX group wants to offer the best service and care for its customers and ensure the safety of the medicines that reach patients. These core principles shape the entire company. For this reason, PHOENIX group has been working diligently and pro-actively on the implementation of the EU Falsified Medicines Directive across all its business areas, so that they are fully compliant with the legislation in all markets. “Our goal was to reach legal compliance on the reporting date of February 9, 2019”, says Karin Bischof-Arden, who was part of the serialization project at PHOENIX group.
However, FMD also significantly challenged PHOENIX group as all its business areas were affected by the directive. In particular, the company’s FMD requirements for wholesale and 3PL activities were challenging to fulfill. “As a wholesaler we only have to scan a small portion of the products which we distribute further”, explains Bischof-Arden, “moreover, common solutions for manufacturers aim at tracing the serial numbers’ life cycle. In our case, however, the serial number of a product appears the first time it is scanned, and we only need to know or update its current status.” Thus, a lean and efficient solution had to be found, which allows capture of the current status of those medicines that a wholesaler has to scan according to FMD.
Movilitas.Cloud proved to be the right solution. It is not only connected to the European Medicines Verification System (EMVS) and to all 32 National Medicine Verification Systems (NMVSs) in Europe, but also acts as a connection between PHOENIX’s systems and the systems of partners. Thus, PHOENIX can decide whether the serialised information of a product is routed to the NMVSs or to the repository of its partners after scanning, or to both. In addition, this solution combines features of a stand-alone (Movilitas.Mobile) and an ERP integrated solution (Wholesaler API). The stand-alone solution’s features enable the scanning of pack codes with mobile devices. The pack coding is based on GS1 standards and includes the GTIN, lot, expiration date, and serial number. The ERP integration allows PHOENIX group to maintain scanning processes that already existed before FMD. In this case Movilitas.Cloud operates in the background and routes all serialised information to the NMVSs or partner systems.
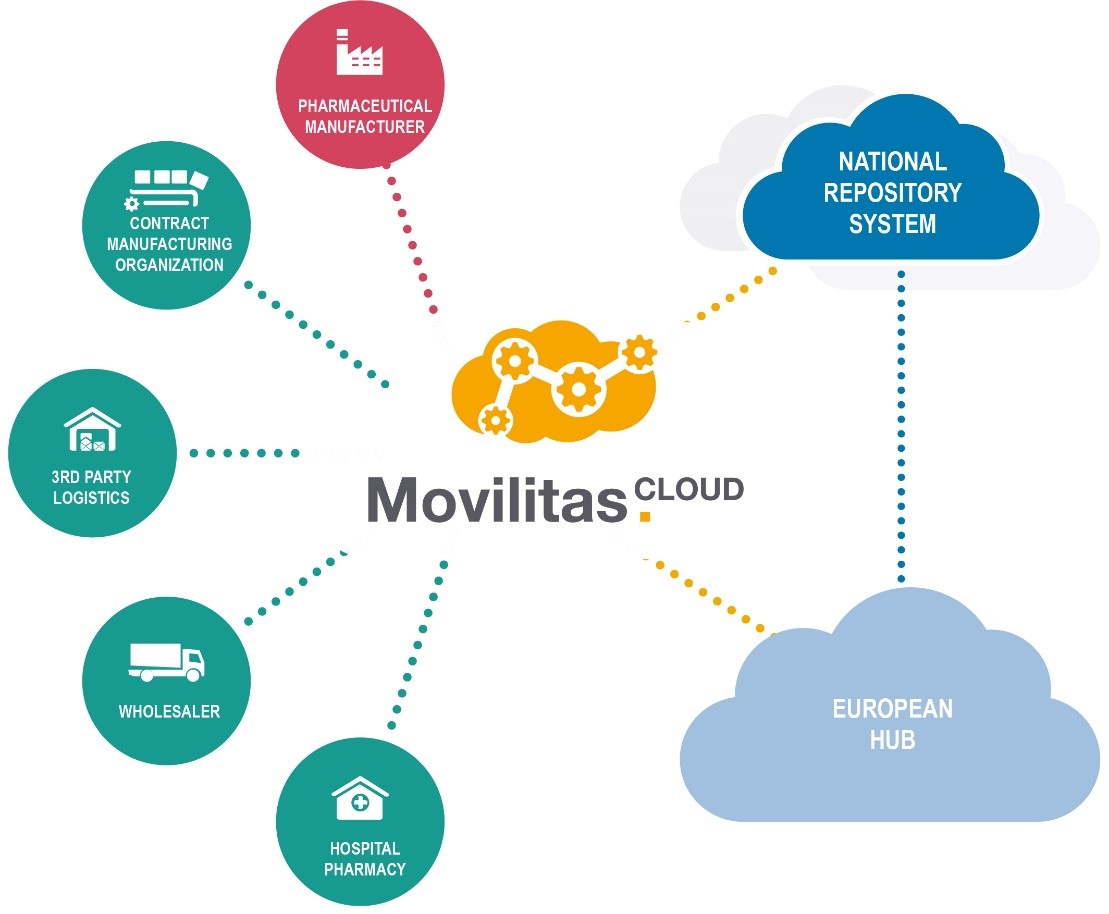
Figure 2 Movilitas Cloud Ecossystem
Another key benefit is that the solution provides real-time feedback on the status of a pack - which is of particular importance for wholesalers as it allows them to immediately trigger required actions and lets them know which pack caused issues. With regard to 3PL activities, PHOENIX group needed to know how many scans they perform for a particular MAH. The Movilitas.Cloud solution also entails detailed reporting capabilities, including not only the number of scans but also a quality report.

Figure 3 PHOENIX group's FMD solution by Movilitas
The role of GS1 standards for FMD
GS1 standards for product identification and labelling are crucial for the successful implementation of such a project. Varying pack codes would not have been sustainable as it would be impossible to scan all the different coding and receive serialised information in such a manner that the PHOENIX systems can interpret the data correctly and work with it. Thus, PHOENIX group relies on manufacturers’ compliance to GS1 standards.
Current situation
FMD is still in its stabilisation period and existing gaps need to be closed to reach a completely secured pharmaceutical supply chain across Europe. Many medicines are not correctly coded yet, a lot of data needs to be uploaded to the EMVS, and process optimisation is required in many areas. However, with the right FMD solution in place, these steps are achievable.
About the Author
Marco Steinkamp
Movilitas Consulting GmbH
Global Sales Director Track & Trace

Marco Steinkamp studied Logistics at Fontys University of Applied Science in Venlo (NL). Afterwards he became an employee at British American Tobacco in 2005 and worked here for 10+ years. For six years he was a member of local and later also the global Supply Chain organisation within BAT. For four years he worked in the Global Track & Trace Program in various roles on local and global level e.g. being the responsible Project Manager in the Logistics as well as afterwards in the Manufacturing environment. With this End to End Supply Chain Track & Trace experience and expertise he joined Movilitas Consulting as the European Sales Director for Track & Trace in May 2015. And since 2017, he has been the Global Sales Director for Track & Trace.